Structure and types of papers
AMP publishes different types of papers according to its editorial policy. The following table shows the maximum word length and other characteristics for the published papers:
Table 2. Maximum length admitted for publication, according to section(s).
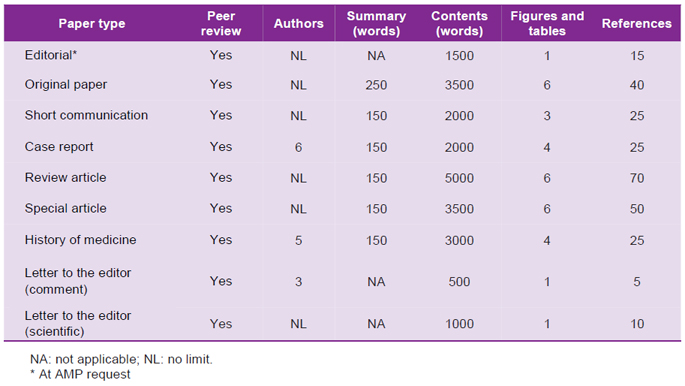
Sections
AMP adheres itself to the Equator-Network initiative (https://www.equator-network.org), and recommends all authors to follow the guidelines adjusting to their study design. From 2021 on, the presentation of the check list included in this initiative will be mandatory, according to the paper type.
Editorial
This is presented under express request from AMP Editorial Committee; its contents must refer to a specific topic of interest about clinical practice, public health matters, issues related to medical activities or ethics, or AMP management or editorial policy. Editorials must have a specific title.
Original papers
These are products of original scientific research, and they must be related to a topic of interest for AMP. These are mainly studies using a prospective and analytical design, with a sample size that is adequate for the research question.
- Abstract. This is structured including objective(s), materials and methods, results, and conclusions. It must be written in Spanish and English.
- Keywords. These must be two as a minimum and six as a maximum. They must be written in Spanish and English. BIREME “science health descriptors” must be used (http://DeCS.bvs.br/) must be used for Spanish and MeSH from NLM (ncbi.mlm-nih.gov/mesh) for English.
- Introduction. This must be brief, going from general to specific features; it usually encompasses less than 20% of the total length of the paper. It must include what is known (relevant background), what is not known (related with the question of research), and what it is to be done (objectives).
- Materials and Methods. Here the used methodology is described, so that it would allow the study to be reproduced and data quality may be evaluated. It is recommended to review international consensus for presenting papers, according to the study design; i.e. STROBE for observational studies, CONSORT for clinical trials, PRISMA for systematic reviews, SRQR for qualitative studies, and CHEERS for economic evaluations. These tools may be found in equator-network.org. The use of subtitles is recommended, including the following information as appropriate:
- Study design. Here the study design is presented, alongside its dates and the place where it has been performed; describing the relevant issues that may help the reader for understanding the conditions in which the study had taken place and that might be useful in the discussion section.
- Study population. Here the study population is described, together with the selection criteria, sample size calculations and power as appropriate, sampling design and enrolling characteristics. It shows a flow chart describing how participants had been enrolled.
- Variables. Here the main study variables are described (relevant dependent and independent variables), so that the variable to be assessed has been assessed in the most appropriate manner, indicating the validity of the used method with its corresponding references, and also the cutoff points may be included in case categorical variables are used.
- Procedures or interventions. These are precisely described, so that any posterior replication of the study may be feasible. Medicinal plant collection and identification procedures are described when they are used. Pharmaceutical products and chemical compounds are to be properly identified, mentioning their active ingredient(s), generic name(s), dose, and administration route(s).
- Data analysis. Here how data is treated is described, including quality control for data bases, statistical software, p-values deemed as significant, tests used for variable assessment, data on assumption compliance and the ways in which models for multiple variables had been developed in case of use.
- Ethical considerations. This describes the approval by Ethics Committees, obtained authorizations, informed consent/assent, data confidentiality, and result return when appropriate.
- Results. There must be clearly presented, not including opinions or interpretations; unless they should correspond to statistical issues. Tables and texts must have a mention within the text, without repeating the information. These may include subtitles in order to facilitate their interpretation.
- Discussion. Here the main results are presented, which may respond to the study objectives. Results are compared with those from other trials, differences or similarities are presented, and reasons for this are stated. Limitations (bias) may be presented in this section, explaining why such findings do not invalidate the results obtained. Here, clinical implications of all findings are presented in detail, stating their importance for research or public health, and recommendations may be issued. Finally, conclusions summarizing the discussion may be presented, based on obtained results and responding to the study objectives.
Short communications
These are also investigation products; and, due to their objectives, design, or results, they may be published using a format not exceeding 2000 words (see Table 2). These are particularly case series, non-probabilistic descriptive studies or small retrospective studies. They follow the same structure as that of an original article. The summary differs from that of an original article because it must not exceed 150 words, and it is not structured.
Review articles
These products should include a wide literature review, they should belong to the areas of interest of AMP, and they must follow this structure: have a non-structured 150-word summary, keywords, introduction, contents (structured according to the author’s discretion), discussion (including conclusions), and references.
Case reports
These products may include from one to ten cases describing an unusual condition, an atypical presentation of a common condition, unknown adverse events, rare disease associations, cases described for the first time in Peru, new interventions or new uses for drugs. All of these situations must have a clear message or lesson for the medical community. The use of CARE guide is recommended for preparing case reports (www.equator-network.org). These products have the following structure: non-structured summary, keywords, introduction (which generally describes the known facts), case report, discussion (where the learned lessons or contributions from presented cases are featured), and references. Information that may help in identifying patient(s) must not be included. In case photographs where it is unavoidable to show the patient’s face because of his/her condition, an explicit authorization from him/her or his/her legal representative must be included. Figures and photographs must be high quality, and they should be independently presented using .jpg or .tiff archives. A physician must always be the corresponding author, and treating physicians should be amongst other authors.
Special articles
These may be essays, opinion papers, clinical practice guides, systematization papers, research protocols, or experience reports that might be of interest for clinical practice, medical education, healthcare policy or being related to medical professional activity. These articles have the following structure: a not more than 150-word non-structured summary, keywords, introduction, contents (structured according to the author’s discretion), discussion (including conclusions), and references.
History of Medicine
Topics for historical review that are relevant in medical fields will be included. These may also include notes about deceased Peruvian physicians. These pieces do not have a summary, their structure is according to the author’s discretion, and the whole piece may include up to 2000 words.
Letters to the Editor
Scientific Letters. These pieces show systematically obtained results. Generally these are descriptive investigations using small non-probabilistic samples. These pieces may include case series or case reports showing specific results of interest or requiring fast-track publication. Scientific letters can have up to 1000 words and they are presented without a summary. These may also constitute a response to a previously published article in a former issue of AMP.
Comment Letters. In the case of non-scientific letters (describing investigation results), their extension may be up to 5000 words, five references one figure or table, and up to three authors. These letters may be a response to an article published in a former issue of AMP, therefore, extending the peer review process, and they may also correspond to evidence-based opinions about areas related to medical education, healthcare policy, being related to medical professional activity, or complaints related to ethical misconducts with respect to some article published in AMP. Implicated authors have the right to respond in the same or in the following issue. In exceptional cases there might be a rejoinder.








1.png)

.png)




